For healthcare professionals

150 Years of Innovation - About Théa Pharmaceuticals
Théa is a privately owned company solely dedicated to ophthalmology and owned by the Chibret family, based in Clermont-Ferrand, France. The story of Théa’s success begins in the 1870s with Paul Chibret, a military doctor. He was stationed in Tunisia and became interested in trachoma, a disease that would, for him, become a lifelong interest and today forms the background behind the Théa Foundation.
In 1875, Paul Chibret opened a private consultancy in Clermont-Ferrand, Central France.
Read the full story (PDF) Watch the video.
In 1902, Paul’s nephew, Henry Chibret, founded Laboratoires Chibret and the company developed an entire range of eye drops and ointments.
In 1943, Jean Chibret pioneered the introduction of preservatives into ophthalmic solutions and in imposed an expiry date for their use after opening the glass vials. Ironically, Jean’s son, Henri Chibret eliminated the use of preservatives that his own father had introduced in 1996 following the increasing recognition that Preservative-Free eyecare is better for the tear film, ocular surface and structures within the eye. Find out more
In 1996, Henri Chibret launched the first Preservative-Free multidose bottle, the ABAK®. Under the leadership of Henri, and his nephew, Jean-Frederic, Théa innovation has continued,and today Jean-Frederic ensures that innovation continues to flourish under the flagship Open Innovation programme. Read more.

1875
Paul Chibret is at the centre of pioneering scientific work in ophthalmology. He is the first of five generations of the Chibret family who will dedicate their lives to driving innovation, education and professionalism in ophthalmology.

1902
Henry Chibret continues his uncle’s work, founding Laboratoires CHIBRET.
1960s
Henry’s son Jean Chibret opens the world’s biggest documentation centre – the CHIBRET Institute.

1970s
Jacques and Henri Chibret both follow in their father’s footsteps launching BIOPHYSIC MEDICAL and Laboratoires TRANSPHYTO.

1994
Laboratoires Théa is launched to sell TRANSPHYO’s products.

1996
Henri Chibret launches the first Preservative-Free, multi-dose bottle. The ABAK® bottle is a high-security, high-technology dispenser which delivers up to 300 drops through a filter which prevents microbial contamination. This leads the way for Preservative-Free formulations and innovative delivery systems. Today, Théa is still highly passionate about Preservative-Free treatments and is dedicated to sharing knowledge with specialists around the world.
View the video, more information and brochure (PDF).
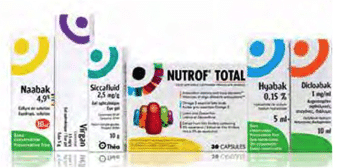
2008
Théa Pharmaceuticals is established. Over the last two decades, under the leadership of Henri Chibret, Théa has achieved numerous world firsts and now offers 30 products across a wide range of key therapy areas in ophthalmology.

2008
Théa Pharmaceuticals UK is established.

2008
Launch of Blephasol®,Blephaclean®, Blephagel®, Nutrof® Total, and Hyabak®.

2010
The first comprehensive moist heat therapy for the management of MGD launched.

2011
Théa acquires Mydriasert® for use in cataract surgery.

2011
Azyter, the 1st 3-day topical antibiotic treatment, is launched.

2011
Tiopex® – 1st low dose timolol gel with no quantifiable systemic absorption.

2012
Théa UK wins Optician Awards 2012 Optical Supplier of the Year Award.

2013
Monopost® – the fist Preservative-Free prostaglandin in single dose format, for the treatment of glaucoma launched along with Aprokam®, the first intracameral antibiotic for prophylaxis of endopthalmitis.

2014
Thealoz® Duo – Unique combination of trehalose and hyaluronic acid without preservatives in an ABAK® bottle for mild to severe dry eye, capitalising on the benefits of Thealoz® and Hyabak®. Also introducing Dexafree®, a Preservative-Free dexamethasone.

2015
The first Théa Birmingham Corneal meeting is held, bringing together the leading authorities in the field of cornea. Watch the latest event in Théa Academy.

2016
Mydrane® – 1st intracameral solution for injection to obtain pupil dilation and intraocular anaesthesia directly in the operating room during cataract surgery.

2017
Blephaclean® is launched into hospitals after achieving CE mark.
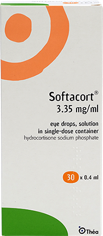
2018
Softacort®, the first Preservative-Free topical hydrocortisone eye drops for use in cases of mild non-infectious conjunctival inflammation launched.

2019
Blephademodex® launch in response to growing need for clinically proven wipe to combat demodex mite infestation.

2019
Fixapost® – 1st Preservative-Free Latanoprost / Timolol Fixed combination in single-dose – In Europe*.
(*with European marketing authorisation)
2019
Théa UK launches national educational series of webinars freely available to Healthcare Professionals.
2021
Théa UK launches the Blepha EyeBag® – a clinically proven medical device for the treatment of blepharitis, styes, chalazion and meibomian gland dysfunction.